Abstract
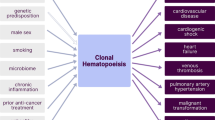
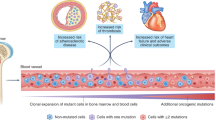
Somatic mutations in hematopoietic stem cells are common with aging and can result in expansion of clones harboring mutations, termed clonal hematopoiesis. This results in an increased risk of blood cancers but has also been linked with chronic inflammatory disease states. In recent years, clonal hematopoiesis has been established to have a causative role in atherogenesis and cardiovascular disease. Additionally, as the effector cells have been identified to be immune cells, there is ongoing interest in assessing whether dysregulated immune function plays a role in other chronic inflammatory conditions such as rheumatologic disease. Here, we summarize current understanding of clonal hematopoiesis with a focus on cardiovascular disease and inflammation while outlining the potential, yet unexplored, relationship between clonal hematopoiesis and autoimmune disease.
Graphical abstract

Similar content being viewed by others
References
Jaiswal S, Fontanillas P, Flannick J et al (2014) Age-related clonal hematopoiesis associated with adverse outcomes. N Engl J Med 371(26):2488–2498
Kakiuchi N, Ogawa S (2021) Clonal expansion in non-cancer tissues. Nat Rev Cancer 21(4):239–256
Weissman IL, Shizuru JA (2008) The origins of the identification and isolation of hematopoietic stem cells, and their capability to induce donor-specific transplantation tolerance and treat autoimmune diseases. Blood 112(9):3543–3553
Osorio FG, Rosendahl Huber A, Oka R et al (2018) Somatic mutations reveal lineage relationships and age-related mutagenesis in human hematopoiesis. Cell Rep 25(9):2308-2316 e2304
Steensma DP, Bejar R, Jaiswal S et al (2015) Clonal hematopoiesis of indeterminate potential and its distinction from myelodysplastic syndromes. Blood 126(1):9–16
Busque L, Mio R, Mattioli J et al (1996) Nonrandom X-inactivation patterns in normal females: lyonization ratios vary with age. Blood 88(1):59–65
Young AL, Challen GA, Birmann BM, Druley TE (2016) Clonal haematopoiesis harbouring AML-associated mutations is ubiquitous in healthy adults. Nat Commun 7:12484
Young AL, Tong RS, Birmann BM, Druley TE (2019) Clonal hematopoiesis and risk of acute myeloid leukemia. Haematologica 104(12):2410–2417
McKerrell T, Park N, Moreno T et al (2015) Leukemia-associated somatic mutations drive distinct patterns of age-related clonal hemopoiesis. Cell Rep 10(8):1239–1245
Bick AG, Weinstock JS, Nandakumar SK et al (2020) Inherited causes of clonal haematopoiesis in 97,691 whole genomes. Nature 586(7831):763–768
Desai P, Mencia-Trinchant N, Savenkov O et al (2018) Somatic mutations precede acute myeloid leukemia years before diagnosis. Nat Med 24(7):1015–1023
Abelson S, Collord G, Ng SWK et al (2018) Prediction of acute myeloid leukaemia risk in healthy individuals. Nature 559(7714):400–404
Genovese G, Kahler AK, Handsaker RE et al (2014) Clonal hematopoiesis and blood-cancer risk inferred from blood DNA sequence. N Engl J Med 371(26):2477–2487
Xie M, Lu C, Wang J et al (2014) Age-related mutations associated with clonal hematopoietic expansion and malignancies. Nat Med 20(12):1472–1478
Dorsheimer L, Assmus B, Rasper T et al (2019) Association of mutations contributing to clonal hematopoiesis with prognosis in chronic ischemic heart failure. JAMA Cardiol 4(1):25–33
Jaiswal S, Natarajan P, Silver AJ et al (2017) Clonal hematopoiesis and risk of atherosclerotic cardiovascular disease. N Engl J Med 377(2):111–121
Yu B, Roberts MB, Raffield LM et al (2021) Supplemental association of clonal hematopoiesis with incident heart failure. J Am Coll Cardiol 78(1):42–52
Yang L, Rau R, Goodell MA (2015) DNMT3A in haematological malignancies. Nat Rev Cancer 15(3):152–165
Perner F, Perner C, Ernst T, Heidel FH (2019) Roles of JAK2 in aging, inflammation, hematopoiesis and malignant transformation. Cells 8(8)
Arends CM, Galan-Sousa J, Hoyer K et al (2018) Hematopoietic lineage distribution and evolutionary dynamics of clonal hematopoiesis. Leukemia 32(9):1908–1919
Carty SA, Gohil M, Banks LB et al (2018) The Loss of TET2 Promotes CD8(+) T cell memory differentiation. J Immunol 200(1):82–91
Ladle BH, Li KP, Phillips MJ et al (2016) De novo DNA methylation by DNA methyltransferase 3a controls early effector CD8+ T-cell fate decisions following activation. Proc Natl Acad Sci USA 113(38):10631–10636
Gamper CJ, Agoston AT, Nelson WG, Powell JD (2009) Identification of DNA methyltransferase 3a as a T cell receptor-induced regulator of Th1 and Th2 differentiation. J Immunol 183(4):2267–2276
Yang R, Qu C, Zhou Y et al (2015) Hydrogen sulfide promotes Tet1- and Tet2-mediated Foxp3 demethylation to drive regulatory T cell differentiation and maintain immune homeostasis. Immunity 43(2):251–263
Ichiyama K, Chen T, Wang X et al (2015) The methylcytosine dioxygenase Tet2 promotes DNA demethylation and activation of cytokine gene expression in T cells. Immunity 42(4):613–626
Fuster JJ, MacLauchlan S, Zuriaga MA et al (2017) Clonal hematopoiesis associated with TET2 deficiency accelerates atherosclerosis development in mice. Science 355(6327):842–847
Zhang Q, Zhao K, Shen Q et al (2015) Tet2 is required to resolve inflammation by recruiting Hdac2 to specifically repress IL-6. Nature 525(7569):389–393
Sano S, Oshima K, Wang Y et al (2018) Tet2-mediated clonal hematopoiesis accelerates heart failure through a mechanism involving the IL-1beta/NLRP3 inflammasome. J Am Coll Cardiol 71(8):875–886
Sano S, Oshima K, Wang Y, Katanasaka Y, Sano M, Walsh K (2018) CRISPR-mediated gene editing to assess the roles of Tet2 and Dnmt3a in clonal hematopoiesis and cardiovascular disease. Circ Res 123(3):335–341
Abplanalp WT, Cremer S, John D et al (2021) Clonal hematopoiesis-driver DNMT3A mutations alter immune cells in heart failure. Circ Res 128(2):216–228
Wang W, Liu W, Fidler T et al (2018) Macrophage inflammation, erythrophagocytosis, and accelerated atherosclerosis in Jak2 (V617F) Mice. Circ Res 123(11):e35–e47
Fidler TP, Xue C, Yalcinkaya M et al (2021) The AIM2 inflammasome exacerbates atherosclerosis in clonal haematopoiesis. Nature 592(7853):296–301
Zekavat SM, Viana-Huete V, Zuriaga MA, et al <em>TP53-</em>mediated clonal hematopoiesis confers increased risk for incident peripheral artery disease. medRxiv. 2021:2021.2008.2022.21262430
Savola P, Lundgren S, Keranen MAI et al (2018) Clonal hematopoiesis in patients with rheumatoid arthritis. Blood Cancer J 8(8):69
Arends CM, Weiss M, Christen F et al (2020) Clonal hematopoiesis in patients with anti-neutrophil cytoplasmic antibody-associated vasculitis. Haematologica 105(6):e264–e267
Niroula A, Sekar A, Murakami MA et al (2021) Distinction of lymphoid and myeloid clonal hematopoiesis. Nat Med 27(11):1921–1927
Ross R (1999) Atherosclerosis--an inflammatory disease. N Engl J Med 340(2):115–126
Ridker PM, Everett BM, Thuren T et al (2017) Antiinflammatory therapy with canakinumab for atherosclerotic disease. N Engl J Med 377(12):1119–1131
Svensson EC, Madar A, Campbell CD, et al (2018) Abstract 15111: TET2-Driven clonal hematopoiesis predicts enhanced response to canakinumab in the CANTOS trial: an exploratory analysis. Circulation 138(Suppl_1):A15111-A15111
Bick AG, Pirruccello JP, Griffin GK et al (2020) Genetic interleukin 6 signaling deficiency attenuates cardiovascular risk in clonal hematopoiesis. Circulation 141(2):124–131
Libby P (2017) Interleukin-1 beta as a target for atherosclerosis therapy: biological basis of CANTOS and beyond. J Am Coll Cardiol 70(18):2278–2289
Hormaechea-Agulla D, Matatall KA, Le DT et al (2021) Chronic infection drives Dnmt3a-loss-of-function clonal hematopoiesis via IFNgamma signaling. Cell Stem Cell 28(8):1428-1442 e1426
Pollard KM, Cauvi DM, Toomey CB, Morris KV, Kono DH (2013) Interferon-gamma and systemic autoimmunity. Discov Med 16(87):123–131
Frostegard J (2005) Atherosclerosis in patients with autoimmune disorders. Arterioscler Thromb Vasc Biol 25(9):1776–1785
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Amancherla serves on an adjudication committee for ACI Clinical. Dr. Bick is on the scientific advisory board of TenSixteen Bio.
Additional information
This article is a contribution to the special issue on: Inflammation in vascular diseases - Guest Editors: Mariana Kaplan & Peter Grayson
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Amancherla, K., Wells, J.A. & Bick, A.G. Clonal hematopoiesis and vascular disease. Semin Immunopathol 44, 303–308 (2022). https://doi.org/10.1007/s00281-022-00913-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00281-022-00913-z