Abstract
Purpose
S-warfarin is used to phenotype cytochrome P450 (CYP) 2C9 activity. This study evaluated S-warfarin limited sampling strategy with a population pharmacokinetic (PK) approach to estimate CYP2C9 activity in healthy adults.
Methods
In 6 previously published studies, a single oral dose of warfarin 10 mg was administered alone or with a CYP2C9 inducer to 100 healthy adults. S-warfarin concentrations were obtained from adults during conditions when subjects were not on any prescribed medications. A population PK model was developed using non-linear mixed effects modeling. Limited sampling models (LSMs) using single- or 2-timepoint concentrations were compared with full PK profiles from intense sampling using empiric Bayesian post hoc estimations of S-warfarin AUC derived from the population PK model. Preset criterion for LSM selection and validation were a correlation coefficient (R2) >0.9, relative percent mean prediction error (%MPE) >−5 to <5%, relative percent mean absolute error (%MAE) ≤ 10%, and relative percent root mean squared error (%RMSE) ≤ 15%.
Results
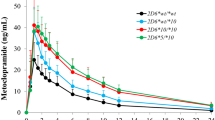
S-warfarin concentrations (n=2540) were well described with a two-compartment model. Mean apparent oral clearance was 0.56 L/hr and volume of distribution was 35.5 L. Clearance decreased 33% with the CYP2C9 *3 allele and increased 42% with lopinavir/ritonavir co-administration. During CYP2C9 constitutive conditions, LSMs at 48 hr and at 72 hr as well as 2-timepoint LSMs were within acceptable limits for R2, %MPE, %MAE, and %RMSE. During CYP2C9 induction, S-warfarin LSMs had unacceptable %MPE, %MAE, and %RMSE.
Conclusions
Phenotyping studies with S-warfarin in healthy subjects can utilize a single- and/or a 2-timepoint LSM with a population PK approach to estimate constitutive CYP2C9 activity.


Similar content being viewed by others
References
Rettie AE, Korzekwa KR, Kunze KL, Lawrence RF, Eddy AC, Aoyama T, Gelboin HV, Gonzalez FJ, Trager WF (1992) Hydroxylation of warfarin by human cDNA-expressed cytochrome P-450: a role for P-4502C9 in the etiology of (S)-warfarin-drug interactions. Chem Res Toxicol 5(1):54–59
Veronese ME, Mackenzie PI, Doecke CJ, McManus ME, Miners JO, Birkett DJ (1991) Tolbutamide and phenytoin hydroxylations by cDNA-expressed human liver cytochrome P4502C9. Biochem Biophys Res Commun 175(3):1112–1118
Zanger UM, Turpeinen M, Klein K, Schwab M (2008) Functional pharmacogenetics/genomics of human cytochromes P450 involved in drug biotransformation. Anal Bioanal Chem 392(6):1093–1108
Food and Drug Adminstration. Drug development and drug interactions: table of substrates, inhibitors and inducers. Available from: https://www.fda.gov/drugs/developmentapprovalprocess/developmentresources/druginteractionslabeling/ucm093664.htm (Accessed 05 January 2021).
Tucker GT, Houston JB, Huang SM (2001) Optimizing drug development: strategies to assess drug metabolism/transporter interaction potential--towards a consensus. Br J Clin Pharmacol 52(1):107–117
Kim JS, Nafziger AN, Gaedigk A, Dickmann LJ, Rettie AE, Bertino JS Jr (2001) Effects of oral vitamin K on S- and R-warfarin pharmacokinetics and pharmacodynamics: enhanced safety of warfarin as a CYP2C9 probe. J Clin Pharmacol 41(7):715–722
Ma JD, Nafziger AN, Kashuba AD, Kim MJ, Gaedigk A, Rowland E, Kim JS, Bertino JS Jr (2004) Limited sampling strategy of S-warfarin concentrations, but not warfarin S/R ratios, accurately predicts S-warfarin AUC during baseline and inhibition in CYP2C9 extensive metabolizers. J Clin Pharmacol 44(6):570–576
Chang AT, Bertino JS Jr, Nafziger AN, Kashuba AD, Turpault S, Lewis LD, Ma JD (2016) S-Warfarin limited sampling models to estimate area under the concentration versus time curve for cytochrome P450 2C9 baseline activity and after induction. Ther Drug Monit 38(3):383–387
Wu JC, Nafziger AN, Bertino JS Jr, Ma JD (2012) Limitations of S-warfarin truncated area under the concentration-time curve to predict cytochrome P450 2c9 activity. Drug Metab Lett 6(2):94–101
Burger D, Ewings F, Kabamba D, L’Homme R, Mulenga V, Kankasa C, Thomason M, Gibb DM, Chintu C, Walker AS (2010) Limited sampling models to predict the pharmacokinetics of nevirapine, stavudine, and lamivudine in HIV-infected children treated with pediatric fixed-dose combination tablets. Ther Drug Monit 32(3):369–372
Ting LS, Villeneuve E, Ensom MH (2006) Beyond cyclosporine: a systematic review of limited sampling strategies for other immunosuppressants. Ther Drug Monit 28(3):419–430
van Rijn SP, Zuur MA, van Altena R, Akkerman OW, Proost JH, de Lange WC, Kerstjens HA, Touw DJ, van der Werf TS, Kosterink JG, Alffenaar JW (2017) Pharmacokinetic modeling and limited sampling strategies based on healthy volunteers for monitoring of ertapenem in patients with multidrug-resistant tuberculosis. Antimicrob Agents Chemother 61(4)
David O, Johnston A (2000) Limited sampling strategies. Clin Pharmacokinet 39(4):311–313
David OJ, Johnston A (2001) Limited sampling strategies for estimating cyclosporin area under the concentration-time curve: review of current algorithms. Ther Drug Monit 23(2):100–114
Yang J, Patel M, Nikanjam M, Capparelli EV, Tsunoda SM, Greenberg HE, Penzak SR, Aubrey Stoch S, Bertino JS Jr, Nafziger AN, Ma JD (2018) Midazolam single time point concentrations to estimate exposure and cytochrome P450 (CYP) 3A constitutive activity utilizing limited sampling strategy with a population pharmacokinetic approach. J Clin Pharmacol 58(9):1205–1213
Johnson BM, Song IH, Adkison KK, Borland J, Fang L, Lou Y, Berrey MM, Nafziger AN, Piscitelli SC, Bertino JS Jr (2006) Evaluation of the drug interaction potential of aplaviroc, a novel human immunodeficiency virus entry inhibitor, using a modified cooperstown 5 + 1 cocktail. J Clin Pharmacol 46(5):577–587
Lee, L.S., Effect of azithromycin, levofloxacin, and telithromycin on the activities of cytochrome P450 1A2, 2C9, 2C19, 2D6, 3A, N-acetyltransferase-2, and xanthine oxidase as assessed by the Cooperstown 5+1 Cocktail in 45th Interscience Conference on Antimicrobial Agents and Chemotherapy. 2005: Washington DC
Ma JD, Nafziger AN, Villano SA, Gaedigk A, Bertino JS Jr (2006) Maribavir pharmacokinetics and the effects of multiple-dose maribavir on cytochrome P450 (CYP) 1A2, CYP 2C9, CYP 2C19, CYP 2D6, CYP 3A, N-acetyltransferase-2, and xanthine oxidase activities in healthy adults. Antimicrob Agents Chemother 50(4):1130–1135
Shelepova T, Nafziger AN, Victory J, Kashuba AD, Rowland E, Zhang Y, Sellers E, Kearns G, Leeder JS, Gaedigk A, Bertino JS Jr (2005) Effect of a triphasic oral contraceptive on drug-metabolizing enzyme activity as measured by the validated Cooperstown 5+1 cocktail. J Clin Pharmacol 45(12):1413–1421
Turpault S, Brian W, Van Horn R, Santoni A, Poitiers F, Donazzolo Y, Boulenc X (2009) Pharmacokinetic assessment of a five-probe cocktail for CYPs 1A2, 2C9, 2C19, 2D6 and 3A. Br J Clin Pharmacol 68(6):928–935
Yeh RF, Gaver VE, Patterson KB, Rezk NL, Baxter-Meheux F, Blake MJ, Eron JJ Jr, Klein CE, Rublein JC, Kashuba AD (2006) Lopinavir/ritonavir induces the hepatic activity of cytochrome P450 enzymes CYP2C9, CYP2C19, and CYP1A2 but inhibits the hepatic and intestinal activity of CYP3A as measured by a phenotyping drug cocktail in healthy volunteers. J Acquir Immune Defic Syndr 42(1):52–60
Lim ML, Min SS, Eron JJ, Bertz RJ, Robinson M, Gaedigk A, Kashuba AD (2004) Coadministration of lopinavir/ritonavir and phenytoin results in two-way drug interaction through cytochrome P-450 induction. J Acquir Immune Defic Syndr 36(5):1034–1040
Karnes JH, Rettie AE, Somogyi AA, Huddart R, Fohner AE, Formea CM, Lee MTM, Llerena A, Whirl-Carrillo M, Klein TE, Phillips EJ, Mintzer S, Gaedigk A, Caudle KE, Callaghan JT (2021) Clinical Pharmacogenetics Implementation Consortium (CPIC) guideline for CYP2C9 and HLA-B genotypes and phenytoin dosing: 2020 update. Clin Pharmacol Ther 109(2):302–309
Johnson JA, Gong L, Whirl-Carrillo M, Gage BF, Scott SA, Stein CM, Anderson JL, Kimmel SE, Lee MT, Pirmohamed M, Wadelius M, Klein TE, Altman RB, Clinical Pharmacogenetics C (2011) Implementation, Clinical Pharmacogenetics Implementation Consortium guidelines for CYP2C9 and VKORC1 genotypes and warfarin dosing. Clin Pharmacol Ther 90(4):625–629
Kamali F, Khan TI, King BP, Frearson R, Kesteven P, Wood P, Daly AK, Wynne H (2004) Contribution of age, body size, and CYP2C9 genotype to anticoagulant response to warfarin. Clin Pharmacol Ther 75(3):204–212
Lindh JD, Holm L, Andersson ML, Rane A (2009) Influence of CYP2C9 genotype on warfarin dose requirements--a systematic review and meta-analysis. Eur J Clin Pharmacol 65(4):365–375
Arab-Alameddine M, Di Iulio J, Buclin T, Rotger M, Lubomirov R, Cavassini M, Fayet A, Decosterd LA, Eap CB, Biollaz J, Telenti A, Csajka C, H.I.V.Cohort Study (2009) Swiss, Pharmacogenetics-based population pharmacokinetic analysis of efavirenz in HIV-1-infected individuals. Clin Pharmacol Ther 85(5):485–494
Sheiner LB, Beal SL (1981) Some suggestions for measuring predictive performance. J Pharmacokinet Biopharm 9:503–512
Mahmood I (2003) Center specificity in the limited sampling model (LSM): can the LSM developed from healthy subjects be extended to disease states? Int J Clin Pharmacol Ther 41(11):517–523
Chaobal HN, Kharasch ED (2005) Single-point sampling for assessment of constitutive, induced, and inhibited cytochrome P450 3A activity with alfentanil or midazolam. Clin Pharmacol Ther 78(5):529–539
Higashi MK, Veenstra DL, Kondo LM, Wittkowsky AK, Srinouanprachanh SL, Farin FM, Rettie AE (2002) Association between CYP2C9 genetic variants and anticoagulation-related outcomes during warfarin therapy. JAMA 287(13):1690–1698
Sconce EA, Khan TI, Wynne HA, Avery P, Monkhouse L, King BP, Wood P, Kesteven P, Daly AK, Kamali F (2005) The impact of CYP2C9 and VKORC1 genetic polymorphism and patient characteristics upon warfarin dose requirements: proposal for a new dosing regimen. Blood 106(7):2329–2333
Scordo MG, Pengo V, Spina E, Dahl ML, Gusella M, Padrini R (2002) Influence of CYP2C9 and CYP2C19 genetic polymorphisms on warfarin maintenance dose and metabolic clearance. Clin Pharmacol Ther 72(6):702–710
Lee CR, Goldstein JA, Pieper JA (2002) Cytochrome P450 2C9 polymorphisms: a comprehensive review of the in-vitro and human data. Pharmacogenetics 12(3):251–263
D’Andrea G, D’Ambrosio RL, Di Perna P, Chetta M, Santacroce R, Brancaccio V, Grandone E, Margaglione M (2005) A polymorphism in the VKORC1 gene is associated with an interindividual variability in the dose-anticoagulant effect of warfarin. Blood 105(2):645–649
Funding
Dr. Tran was a post-doctoral fellow with funding supported by the University of North Carolina at Chapel Hill and Pharmaceutical Product Development. Funding support was provided by a Research in Pediatric and Developmental Pharmacology NIH grant (1U54HD090259-01 to E.V.C.)
Author information
Authors and Affiliations
Contributions
LT, MN, EVC, and JDM contributed to the study design. LT, MN, and JDM were involved in data collection. LT, MN, EVC, JSB, ANN, and JDM contributed to data interpretation and statistical analysis. All authors reviewed, edited, and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 397 kb)
Rights and permissions
About this article
Cite this article
Tran, L., Nikanjam, M., Capparelli, E.V. et al. S-warfarin limited sampling strategy with a population pharmacokinetic approach to estimate exposure and cytochrome P450 (CYP) 2C9 activity in healthy adults. Eur J Clin Pharmacol 77, 1349–1356 (2021). https://doi.org/10.1007/s00228-021-03123-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-021-03123-y