Abstract
Summary
In the Diabetes Prevention Program Outcome Study (DPPOS), a cohort at high risk of diabetes, randomization to intensive lifestyle intervention or metformin, both associated with weight loss, did not have long-term negative effects on BMD compared with the placebo group. Potential positive effects of metformin on bone warrant further investigation.
Introduction
Randomization to lifestyle intervention (ILS) or metformin in the Diabetes Prevention Program (DPP) resulted in weight loss and reduced progression to diabetes. Weight loss is associated with reduced bone mineral density (BMD), but the long-term effects of these interventions on BMD are unknown. In the DPP Outcome Study (DPPOS), we determined if randomization to ILS or metformin, compared with placebo, was associated with differences in BMD approximately 16 years later.
Methods
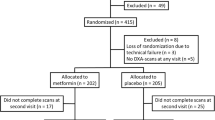
Of 3234 DPP participants, 2779 continued in DPPOS and were offered ILS in group format. Those randomized to metformin were offered unmasked metformin. At DPPOS year 12, 1367 participants had dual-energy X-ray absorptiometry scans. BMD in metformin and ILS groups was compared to placebo using sex-specific linear regression models, adjusted for age, race/ethnicity, and weight and weight-bearing activity at DPP baseline.
Results
At DPPOS year 12, mean age was 66.5 (±9.5) years. Femoral neck BMD was similar in the ILS and placebo groups in men (difference = −0.021 g/cm2, 95%CI (−0.063, 0.021)) and in women (+0.014 g/cm2, 95%CI (−0.014, 0.042)). Femoral neck BMD was higher in the metformin compared to placebo group although not statistically different in men (+0.017 g/cm2, 95% CI (−0.023, 0.058)) and in women (+0.019 g/cm2, 95% CI (−0.009, 0.047)). Prevalence of osteoporosis was low and similar across treatment groups in men (0.9%; p=0.745) and women (2.4%; p=0.466).
Conclusion
In a cohort at high risk of diabetes, lifestyle intervention or metformin did not appear to have long-term negative effects on BMD. Potential positive effects of metformin on bone warrant further research.



Similar content being viewed by others
Availability of data and material
Some or all data generated or analyzed during this study are available in the NIDDK data repository.
References
Knowler WC, Barrett-Connor E, Fowler SE, Hamman RF, Lachin JM, Walker EA, Nathan DM (2002) Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med 346:393–403
Group DPPR (2012) Long-term safety, tolerability, and weight loss associated with metformin in the Diabetes Prevention Program Outcomes Study. Diabetes Care 35:731–737
Ensrud KE, Fullman RL, Barrett-Connor E, Cauley JA, Stefanick ML, Fink HA, Lewis CE, Orwoll E (2005) Voluntary weight reduction in older men increases hip bone loss: the osteoporotic fractures in men study. J Clin Endocrinol Metab 90:1998–2004
Hannan MT, Felson DT, Dawson-Hughes B, Tucker KL, Cupples LA, Wilson PW, Kiel DP (2000) Risk factors for longitudinal bone loss in elderly men and women: the Framingham Osteoporosis Study. J Bone Miner Res 15:710–720
Schwartz AV, Johnson KC, Kahn SE, Shepherd JA, Nevitt MC, Peters AL, Walkup MP, Hodges A, Williams CC, Bray GA (2012) Effect of one year of an intentional weight loss intervention on bone mineral density in type 2 diabetes: results from the Look AHEAD randomized trial. J Bone Miner Res 27:619–627
Lipkin EW, Schwartz AV, Anderson AM, Davis C, Johnson KC, Gregg EW, Bray GA, Berkowitz R, Peters AL, Hodges A, Lewis C, Kahn SE, the Look AHEAD Research Group (2014) The Look AHEAD Trial: bone loss at 4-year follow-up in type 2 diabetes. Diabetes Care 37:2822–2829
McCarthy AD, Cortizo AM, Sedlinsky C (2016) Metformin revisited: does this regulator of AMP-activated protein kinase secondarily affect bone metabolism and prevent diabetic osteopathy. World J Diabetes 7:122–133
Knowler WC, Fowler SE, Hamman RF, Christophi CA, Hoffman HJ, Brenneman AT, Brown-Friday JO, Goldberg R, Venditti E, Nathan DM (2009) 10-year follow-up of diabetes incidence and weight loss in the Diabetes Prevention Program Outcomes Study. Lancet 374:1677–1686
Hui SL, Gao S, Zhou XH, Johnston CC Jr, Lu Y, Gluer CC, Grampp S, Genant H (1997) Universal standardization of bone density measurements: a method with optimal properties for calibration among several instruments. J Bone Miner Res 12:1463–1470
Lu Y, Fuerst T, Hui S, Genant HK (2001) Standardization of bone mineral density at femoral neck, trochanter and Ward's triangle. Osteoporos Int 12:438–444
Kanis JA, Bianchi G, Bilezikian JP, Kaufman JM, Khosla S, Orwoll E, Seeman E (2011) Towards a diagnostic and therapeutic consensus in male osteoporosis. Osteoporos Int 22:2789–2798
Kriska AM, Knowler WC, LaPorte RE, Drash AL, Wing RR, Blair SN, Bennett PH, Kuller LH (1990) Development of questionnaire to examine relationship of physical activity and diabetes in Pima Indians. Diabetes Care 13:401–411
Kriska AM, Rockette-Wagner B, Edelstein SL, Bray GA, Delahanty LM, Hoskin MA, Horton ES, Venditti EM, Knowler WC, DPP Research Group (2021) The impact of physical activity on the prevention of type 2 diabetes: evidence and lessons learned from the diabetes prevention program, a long-standing clinical trial incorporating subjective and objective activity measures. Diabetes Care 44:43–49
Zibellini J, Seimon RV, Lee CM, Gibson AA, Hsu MS, Shapses SA, Nguyen TV, Sainsbury A (2015) Does diet-induced weight loss lead to bone loss in overweight or obese adults? A systematic review and meta-analysis of clinical trials. J Bone Miner Res 30:2168–2178
Venditti EM, Bray GA, Carrion-Petersen ML, Delahanty LM, Edelstein SL, Hamman RF, Hoskin MA, Knowler WC, Ma Y, Diabetes Prevention Program Research Group (2008) First versus repeat treatment with a lifestyle intervention program: attendance and weight loss outcomes. Int J Obes 32:1537–1544
Shojaa M, Von Stengel S, Schoene D et al (2020) Effect of exercise training on bone mineral density in post-menopausal women: a systematic review and meta-analysis of intervention studies. Front Physiol 11:652
Pinheiro MB, Oliveira J, Bauman A, Fairhall N, Kwok W, Sherrington C (2020) Evidence on physical activity and osteoporosis prevention for people aged 65+ years: a systematic review to inform the WHO guidelines on physical activity and sedentary behaviour. Int J Behav Nutr Phys Act 17:150
Yarizadeh H, Asadi S, Baharlooi H, Setayesh L, Kakavandi NR, Hambly C, Djafarian K, Mirzaei K (2021) Beneficial impact of exercise on bone mass in individuals under calorie restriction: a systematic review and Meta-analysis of randomized clinical trials. Crit Rev Food Sci Nutr 61:553–565
Villareal DT, Aguirre L, Gurney AB, Waters DL, Sinacore DR, Colombo E, Armamento-Villareal R, Qualls C (2017) Aerobic or resistance exercise, or both, in dieting obese older adults. N Engl J Med 376:1943–1955
Ryan DH, Espeland MA, Foster GD, Haffner SM, Hubbard VS, Johnson KC, Kahn SE, Knowler WC, Yanovski SZ (2003) Look AHEAD (Action for Health in Diabetes): design and methods for a clinical trial of weight loss for the prevention of cardiovascular disease in type 2 diabetes. Control Clin Trials 24:610–628
Johnson KCLC, Womack C, Garcia KR, Wagenknecht LE, Pownall HJ, Horton ES, Pi-Sunyer X, Gregg E, Schwartz AV (2016) The effect of intentional weight loss on fracture risk in diabetics: results from the look AHEAD Clinical Trial. J Bone Miner Res 65:LB-80
Wang C, Li H, Chen SG, He JW, Sheng CJ, Cheng XY, Qu S, Wang KS, Lu ML, Yu YC (2012) The skeletal effects of thiazolidinedione and metformin on insulin-resistant mice. J Bone Miner Metab 30:630–637
Lecka-Czernik B (2017) Diabetes, bone and glucose lowering agents – Basic biology. Diabetologia 60:1163–1169
Starup-Linde J, Gregersen S, Frost M, Vestergaard P (2016) Use of glucose-lowering drugs and risk of fracture in patients with type 2 diabetes. Bone 95:136–142
Kahn SE, Zinman B, Lachin JM, Haffner SM, Herman WH, Holman RR, Kravitz BG, Yu D, Heise MA, Aftring RP, Viberti G, for the A Diabetes Outcome Progression Trial (ADOPT) Study Group (2008) Rosiglitazone associated fractures in type 2 diabetes: an analysis from ADOPT. Diabetes Care 31:845–851
de Liefde II, van der Klift M, de Laet CE, van Daele PL, Hofman A, Pols HA (2005) Bone mineral density and fracture risk in type-2 diabetes mellitus: the Rotterdam Study. Osteoporos Int 16:1713–1720
Napoli N, Strotmeyer ES, Ensrud KE, Sellmeyer DE, Bauer DC, Hoffman AR, Dam TTL, Barrett-Connor E, Palermo L, Orwoll ES, Cummings SR, Black DM, Schwartz AV (2014) Fracture risk in diabetic elderly men: the MrOS study. Diabetologia 57:2057–2065
Napoli N, Conte C, Eastell R, Ewing SK, Bauer DC, Strotmeyer ES, Black DM, Samelson EJ, Vittinghoff E, Schwartz AV (2020) Bone turnover markers do not predict fracture risk in type 2 diabetes. J Bone Miner Res 35:2363–2371
Billington EO, Grey A, Bolland MJ (2015) The effect of thiazolidinediones on bone mineral density and bone turnover: systematic review and meta-analysis. Diabetologia 58:2238–2246
Meier C, Schwartz AV, Egger A, Lecka-Czernik B (2016) Effects of diabetes drugs on the skeleton. Bone 82:93–100
Mabilleau G, Bouvard B (2020) Update on: effects of anti-diabetic drugs on bone metabolism. Expert Rev Endocrinol Metab 15:415–430
Acknowledgements
The Research Group gratefully acknowledges the commitment and dedication of the participants of the DPP and DPPOS.
Funding
Research reported in this publication was supported by the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) of the National Institutes of Health (NIH) under Award Number U01 DK048489, by providing funding during DPP and DPPOS to the clinical centers and the Coordinating Center for the design and conduct of the study, and collection, management, analysis, and interpretation of the data. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. The Southwestern American Indian Centers were supported directly by the NIDDK, including its Intramural Research Program, and the Indian Health Service. The General Clinical Research Center Program, National Center for Research Resources, and the Department of Veterans Affairs supported data collection at many of the clinical centers. Funding was also provided by the National Institute of Child Health and Human Development, the National Institute on Aging, the National Eye Institute, the National Heart Lung and Blood Institute, the National Cancer Institute, the Office of Research on Women’s Health, the National Institute on Minority Health and Health Disparities, the Centers for Disease Control and Prevention, and the American Diabetes Association. Merck KGaA provides medication for DPPOS. DPP/DPPOS have also received donated materials from Bristol-Myers Squibb, Parke-Davis, and LifeScan Inc. LifeScan Inc., Health O Meter, Hoechst Marion Roussel, Inc., Merck-Medco Managed Care, Inc., Merck and Co., Nike Sports Marketing, Slim Fast Foods Co., and Quaker Oats Co. donated materials, equipment, or medicines for concomitant conditions. McKesson BioServices Corp., Matthews Media Group, Inc., and the Henry M. Jackson Foundation provided support services under subcontract with the Coordinating Center. The sponsor of this study was represented on the Steering Committee and played a part in study design, how the study was done, and publication. The opinions expressed are those of the study group and do not necessarily reflect the views of the funding agencies.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Institutional review boards at all sites approved the DPP and DPPOS protocols and informed consent procedures.
Consent to participate
Written informed consent was obtained from all individual participants included in the study.
Conflicts of interest
None.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
A complete list of the DPPOS Centers, investigators, and staff can be found in the Appendix ESM.
Rights and permissions
About this article
Cite this article
Schwartz, A.V., Pan, Q., Aroda, V.R. et al. Long-term effects of lifestyle and metformin interventions in DPP on bone density. Osteoporos Int 32, 2279–2287 (2021). https://doi.org/10.1007/s00198-021-05989-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-021-05989-1